Table of Contents
Overview of Domestic CAR-T Landscape

Domestic CAR-T related stocks are now categorized into two types. The first are companies close to approval and revenue, while the second are those with excellent platforms but still in development stages.
Recently, as market interest in in vivo CAR-T has increased, it is evident that even within the same cell therapy theme, the characteristics of technology and the speed of commercialization greatly vary. These changes offer investors a variety of options.

The following is a condensed and reorganized summary of key points.
The CAR-T company closest to commercialization is Curexcell. GC Cell, including introduced items, is a rapid revenue candidate. Apclon, aiming for CAR-T commercialization as a latecomer, is also gaining attention. Apclon has established itself as a leading company in in vivo CAR-T, while GC Cell and Paxel Bio are considered next-generation expansion platform stocks.

Currently, the domestic CAR-T market is led by Curexcell and Apclon. GC Cell focuses on CAR-NK and commercialization of introduced items, while Paxel Bio is establishing itself as a next-generation modified platform. This distinction provides a practical approach to understanding the market.
Recommended Reads for Bio Stock Investment
Curexcell Leading Commercialization

Currently, the number one CAR-T therapy developed in Korea is Curexcell. The company's CD19 CAR-T product Anbalsel (Limkato injection) has already completed the licensing and reimbursement evaluation process, and it has secured a commercial GMP manufacturing facility.
The final results of Phase 2 clinical trials showed a very positive response rate. For this reason, Curexcell is assessed to be the company most likely to receive permission first among domestically developed CAR-T therapies, with expectations for revenue as well.

Curexcell's strengths do not simply stop at the rapid approval stage. It currently has licensing applications and reimbursement procedures underway, along with commercial GMP manufacturing infrastructure. Furthermore, it is planning to expand from autologous CAR-T to allogeneic γδ CAR-T. Efforts are also being made to expand indications to autoimmune diseases like SLE.
Ultimately, Curexcell is not just a simple early-stage technology stock, but it is positioning itself as the representative company with the clearest commercialization potential in the domestic CAR-T field.
Comparison of GC Cell and Apclon
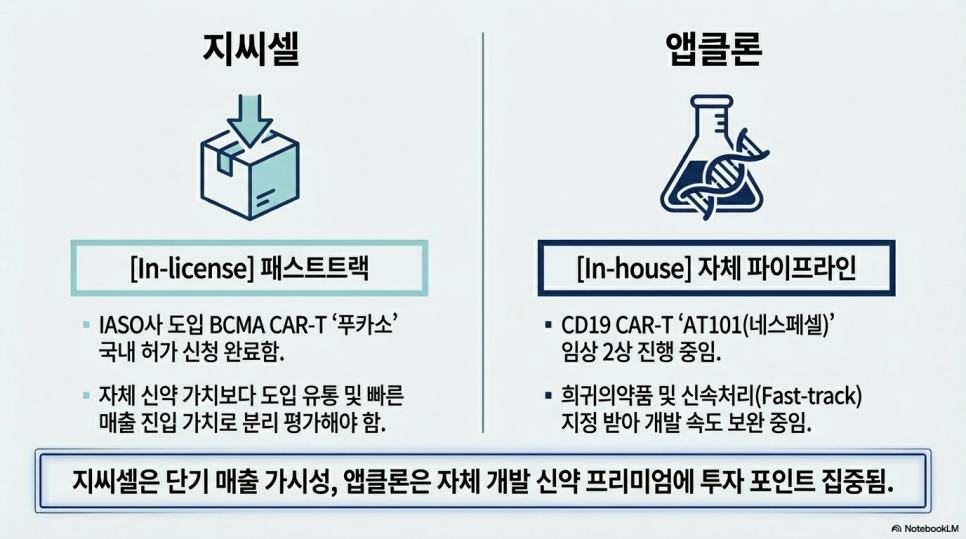
Considering introduced items, GC Cell's commercialization potential is very high. This company has applied for domestic approval for BCMA CAR-T Fucasso at IASO. If the approval is granted, it seems likely to generate revenue relatively quickly. However, it is important to keep in mind that this position is closer to the commercialization value of introduced products than the value of self-developed new drugs.
On the other hand, Apclon is gaining attention as a latecomer with its own CAR-T technology. The CD19 CAR-T AT101 (Nespecell) is currently in phase 2 clinical trials and has already received orphan drug and fast-track designations. Although it is still at a pre-licensing stage, making its proximity to commercialization lower than that of Curexcell, it is establishing itself as the most prominent latecomer in the field of self-developed CAR-T.

In summary of the comparison of the three companies:
In terms of commercialization proximity (including introductions), Curexcell and GC Cell show similar levels, while Apclon lags behind. For commercialization proximity (self-developed only), Curexcell leads, followed by Apclon, with Paxel Bio receiving a relatively low rating.
In terms of purity of self-developed CAR-T, Curexcell scores the highest, followed by Apclon, while Paxel Bio receives the lowest ranking.
In conclusion, from an investment perspective, Curexcell appears to have the highest feasibility, while GC Cell seems to have the potential for rapid revenue entry. Apclon is evaluated as a promising candidate for subsequent commercialization.
Core of in vivo CAR-T

Recently, the keyword gaining a lot of attention is in vivo CAR-T. However, currently, there are no companies at the clinical stage among domestic listed companies. Among them, the company that attracts the most attention is Apclon.
Apclon is positioning in vivo CAR-T as a new growth engine, emphasizing CAR design, mRNA, LNP, and targeting technology as key elements. Particularly, they aim for a day-of-hospital-visit injection type CAR-T, which is generating considerable expectations.
However, looking at the current situation critically, Apclon is at the stage of research and platform development, not clinical entry. Therefore, while the company can be regarded as a representative of domestic in vivo CAR-T, it is primarily the anticipation of future technologies rather than actual revenue or approval possibilities that are being reflected.

Curexcell shows more prominent results in the next-generation ex vivo CAR-T field than in vivo CAR-T. By utilizing the OVIS platform, it maximizes the activity of CAR-T and has a structure that enables expansion to allogeneic γδ CAR-T. So if Apclon is regarded as the representative of in vivo CAR-T, Curexcell can be evaluated as the leading player in advanced ex vivo CAR-T. This structure effectively highlights the differences in technological capabilities among the companies.
Next Generation CAR Expansions

Looking into CAR development companies, GC Cell is focusing more on CAR-NK than CAR-T. Paxel Bio is focusing on CAR-MIL and dual CAR-T.
GC Cell is placing more emphasis on the value of its own platform on CAR-NK, separate from CAR-T commercialization.

Paxel Bio is developing technologies including BCMA CAR-MIL, anti-PD-L1 CAR-T, and dual CAR-T. However, these are still in the preclinical and IND preparation stages, so time is needed for commercialization.
Thus, these stocks are better viewed as next-generation CAR platforms rather than immediate approval stocks. Their technological possibilities are intriguing, but the current visibility of revenue is low and the situation is highly volatile.
Conclusion on Domestic CAR-T
Looking at domestic CAR-T related stocks, Curexcell and Apclon are playing significant roles. Among these, Curexcell is noted as the company closest to commercialization, and considering introduced items, GC Cell is also expected to realize rapid sales.
Although Apclon stands out in the in vivo CAR-T field, it is currently in the research stage. The next-generation expansion platforms realistically attracting attention are GC Cell's CAR-NK and Paxel Bio's CAR-MIL and dual CAR-T. Within this flow, the advancement of CAR-T related companies is anticipated.

When evaluated critically, the following rankings emerge.
In terms of proximity to commercialization and approval, Curexcell is in the lead, followed by GC Cell, Apclon, and Paxel Bio. In terms of purity of self-developed CAR-T, Curexcell occupies the top spot, followed by Apclon, with Paxel Bio in last place. In terms of next-generation technological capability, Apclon is the most prominent, followed by Curexcell, Paxel Bio, and GC Cell.
Ultimately, the most important factor in considering domestic CAR-T investments is to clearly distinguish whether the proximity to current approval and revenue is crucial or if future platform premiums take precedence.
Based on this analysis, Curexcell is classified as the representative for commercialization, Apclon as the leader in next-generation technology, GC Cell as a rapid revenue entry based on introductions, and Paxel Bio as an early platform stock.
※ This article is not intended as an investment recommendation but rather for personal record and information sharing purposes. It does not recommend the purchase or sale of specific assets or stocks.
Frequently Asked Questions (FAQ)
Q. How are domestic CAR-T related stocks classified?
Domestic CAR-T related stocks are categorized into companies close to approval and revenue and those in development stages.
Domestic CAR-T related stocks can be broadly classified into two categories. The first are companies close to approval and revenue, while the second are those with excellent platforms but still in development stages. This classification is crucial in providing investors with various options based on commercialization potential and technological advancement levels.
Q. What position does Curexcell hold in the domestic CAR-T market?
Curexcell is the representative company closest to commercialization of self-developed CAR-T therapies in Korea.
Curexcell has developed the CD19 CAR-T product Anbalsel (Limkato injection) and has completed licensing and reimbursement evaluation requests, securing a commercial GMP manufacturing facility. It shows a positive response rate in phase 2 clinical trials and is most likely to receive permission among domestically developed CAR-T therapies. Furthermore, it is attempting to expand indications for autoimmune diseases while planning to extend to allogeneic γδ CAR-T, making it regarded as a company with clear commercialization potential, not just a simple technology stock.
Q. What characteristics and market strategies does GC Cell possess?
GC Cell focuses on the rapid commercialization of introduced items and the CAR-NK platform.
GC Cell has applied for domestic approval of its BCMA CAR-T product Fucasso, which is expected to generate revenue quickly upon approval. However, it leans more towards the commercialization of introduced products than its self-developed new drugs. Technologically, it is enhancing its CAR-NK platform, adopting a strategy to concentrate on CAR-NK values separate from CAR-T commercialization.
Q. What is Apclon's CAR-T technology and market position?
Apclon is a latecomer based on in vivo CAR-T technology and a next-generation expansion platform company.
Apclon is currently in phase 2 clinical trials for its CD19 CAR-T AT101 (Nespecell) and has received orphan drug and fast-track designations. It emphasizes in vivo CAR-T technology as a core focus, with key elements being CAR design, mRNA, LNP, and targeting technology. While aiming for a day-of-hospital-visit injection type CAR-T, it is still in the research and platform development stage, needing time for commercialization. It carries high expectations regarding its technological capabilities as a representative of in vivo CAR-T in Korea.
Q. Who are the next-generation CAR platform companies in Korea and what is their current status?
Paxel Bio is developing CAR-MIL and dual CAR-T, currently in the preclinical stage.
Paxel Bio is working on technologies such as BCMA CAR-MIL, anti-PD-L1 CAR-T, and dual CAR-T, but remains at the preclinical and IND preparation stages. As a result, immediate commercialization is difficult and it is regarded as an early-stage company with technological potential as a next-generation CAR platform. Given the high volatility and uncertainty, a long-term investment approach is recommended.
Q. What considerations should be made when investing in domestic CAR-T related stocks?
It is essential to distinguish between proximity to approval and revenue versus future platform premiums.
In domestic CAR-T investments, it is crucial to clearly differentiate whether to emphasize the current proximity to approval and revenue, or prioritize the future growth potential of next-generation platforms. Curexcell is regarded as the representative for commercialization, GC Cell as a rapid revenue entry stock based on introductions, and Apclon as the leading stock for next-generation technology. Investment strategies may differ depending on the technological capabilities, approval situations, and speeds of market entry of each company.



